
BIGBEAR
PHARMACEUTICAL
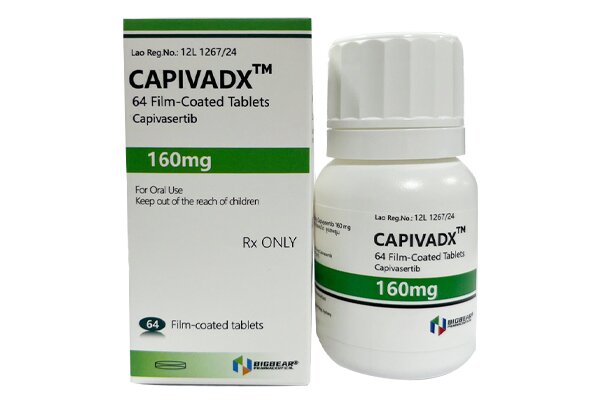
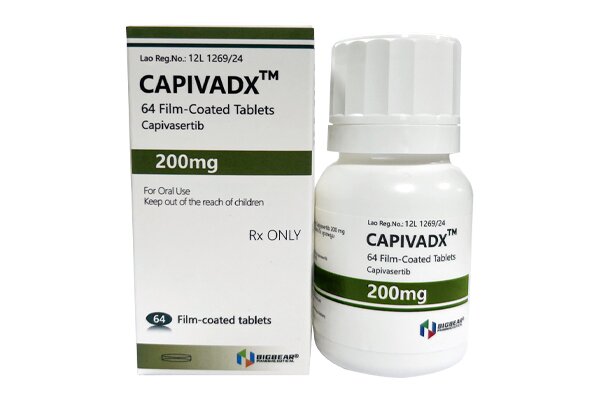
Capivasertib is the first approved highly selective ATP-competitive AKT kinase inhibitor, and is commonly used in combination with fulvestrant.

 Authentic
Authentic Guarantee
Guarantee Fast Delivery
Fast Delivery Privacy
Privacy Capivasertib potently inhibits all three isoforms of AKT1/2/3, blocks aberrant activation of the PI3K/AKT/mTOR signaling pathway, thereby inhibiting tumor cell proliferation, survival and metastasis, and reversing endocrine therapy resistance.
Capivasertib in combination with fulvestrant is indicated for the treatment of adult patients with hormone receptor (HR)-positive, human epidermal growth factor receptor 2 (HER2)-negative, locally advanced or metastatic breast cancer with one or more PIK3CA/AKT1/PTEN alterations, whose disease has progressed following at least one endocrine therapy in the metastatic setting, or who have relapsed during adjuvant therapy or within 12 months after completion of adjuvant therapy.
Prior to use of this product, selection of patients with advanced or metastatic HR‑positive, HER2‑negative breast cancer eligible for treatment should be based on the presence of one or more PIK3CA/AKT1/PTEN alterations in tumor tissue.
For PIK3CA mutations, the mutation status must be determined using a test approved by the National Medical Products Administration or a fully validated assay.
For AKT1/PTEN alterations, the alteration status must be determined using a fully validated assay.
Patients with one or more AKT1/PTEN alterations confirmed by hospital or laboratory testing may receive this product.
AKT1/PTEN alteration status should be tested using a companion diagnostic assay at a facility designated by AstraZeneca Pharmaceuticals Co., Ltd. Treatment may continue only if AKT1/PTEN alterations are confirmed.
Measure fasting glucose (FG) and glycated hemoglobin (HbA1c) prior to initiation of treatment and at regular intervals during treatment.
When used in combination with fulvestrant, the recommended dose of this product is 400 mg orally twice daily (approximately 12 hours apart), with or without food.
The dosing schedule is 4 days of continuous dosing followed by 3 days off per week.
Treatment should be continued until disease progression or unacceptable toxicity occurs.
This product should be swallowed whole.
Do not chew, crush, or break tablets before swallowing.
Do not take any tablets that are broken, cracked, or incomplete.
Dose adjustment is not recommended for patients with mild to moderate renal impairment (creatinine clearance [CLcr] 30–89mL/min).
This product has not been studied in patients with severe renal impairment (CLcr 15–29 mL/min).
Dose adjustment is not recommended for patients with mild hepatic impairment (bilirubin ≤ upper limit of normal [ULN] and AST > ULN, or bilirubin >1–1.5×ULN and AST of any value).
Monitor patients with moderate hepatic impairment (bilirubin > 1.5–3×ULN and AST of any value) for adverse reactions due to potential increased capivasertib exposure.
This product has not been studied in patients with severe hepatic impairment (bilirubin > 3×ULN and AST of any value).
No dose adjustment is required in geriatric patients. Data on the use of this product in patients aged ≥ 75 years are limited.
The safety and efficacy of this product in pediatric patients have not been established.
In this safety population, the most common adverse reactions, including laboratory abnormalities, were diarrhea, skin adverse reactions, random blood glucose elevation, lymphopenia, hemoglobin decrease, fasting blood glucose elevation, nausea and fatigue, leukopenia, triglyceride elevation, neutropenia, creatinine increase, vomiting, and stomatitis.
Severe adverse reactions occurred in patients receiving this product in combination with fulvestrant. The most common severe adverse reactions were skin adverse reactions, diarrhea and infectious pneumonia, vomiting and fever, hyperglycemia, hypersensitivity, fatigue, renal impairment, and second malignant tumors.
Fatal adverse reactions occurred in patients receiving this product in combination with fulvestrant, including sepsis and acute myocardial infarction.
Hypersensitivity to the active substance or to any of the excipients of this product is contraindicated.
Prior to initiation of treatment with this product, measure fasting plasma glucose (FPG) or fasting blood glucose (FBG) and HbA1c, and achieve adequate control of fasting blood glucose.
After initiation of treatment, monitor or self-monitor fasting blood glucose levels on Day 3 or 4 of Weeks 1, 2, 4, 6, and 8; monthly thereafter during treatment; and when clinically indicated.
Monitor HbA1c every 3 months during treatment and when clinically indicated.
Patients with a history of well-controlled type 2 diabetes mellitus may require intensified antihyperglycemic therapy and close monitoring of fasting blood glucose.
For patients who develop hyperglycemia during treatment, monitor fasting blood glucose at least twice weekly (on dosing days and off-dosing days) until levels return to baseline.
During treatment with antihyperglycemic medicinal products, monitor fasting blood glucose at least weekly for 2 months, then every 2 weeks or when clinically indicated.
Consider consultation with a specialist with expertise in the management of hyperglycemia, and consider advising patients with risk factors for or who develop hyperglycemia to perform fasting blood glucose monitoring at home.
Inform patients of the signs and symptoms of hyperglycemia and advise on lifestyle modifications.
For patients with suspected ketoacidosis, immediately interrupt dosing with this product.
If ketoacidosis is confirmed, discontinue this product permanently.
Dosing may be interrupted, reduced, or permanently discontinued based on the severity of hyperglycemia.
Diarrhea may occur in patients receiving this product, which can be severe and lead to dehydration.
Patients should be advised to increase oral fluid intake and initiate antidiarrheal therapy at the first sign of diarrhea during treatment.
Dosing may be interrupted, reduced, or permanently discontinued based on the severity of diarrhea.
Skin adverse reactions, including severe skin adverse reactions such as erythema multiforme (EM), palmar-plantar erythrodysesthesia syndrome, and drug reaction with eosinophilia and systemic symptoms (DRESS), may occur in patients receiving this product.
Monitor patients for signs and symptoms of skin adverse reactions.
Early consultation with a dermatologist is recommended.
Dosing may be interrupted, reduced, or permanently discontinued based on the severity of the skin adverse reaction.
For more detailed drug information, please consult the official package leaflet.
If any issues arise, please contact us immediately.
Email:laosbigbear@gmail.com

Take the medication as soon as you remember. However, if it has been more than 4 hours since your scheduled dose, skip the missed one. Do not take a double dose to make up for it.
Seek immediate emergency medical assistance or contact the Poison Control Helpline.
Grapefruit and grapefruit-containing products may interact with capivasertib, potentially triggering adverse reactions. Refrain from consuming any grapefruit products during treatment.
Certain medications may not be safely used concurrently. Some drugs can alter the blood concentrations of other medications you are taking, which may heighten the risk of side effects or reduce therapeutic efficacy.
Capivasertib may be affected by other medicinal products, including prescription drugs, over-the-counter medications, vitamins, and herbal supplements. Inform your healthcare provider of all medications you are currently using.
Copyright2024@ BIGBEAR All right reserved Bigbear | Bigbear Pharmaceutical | Bigbear Laos



